US Endocrinology, a peer-reviewed journal of research relating to the endocrine system, recently published a focused review on the positive effects of a relatively new combination treatment for obesity. The report is titled, “Sustained-release Naltrexone/Bupropion – A Novel Pharmacologic Approach to Obesity and Food Craving” and is authored by researcher Ken Fujioka.
Naltrexone/Bupropion, a novel anti-obesity combination drug, features bupropion, a reuptake inhibitor that stimulates the release of norepinephrine, and a nicotinic acetylcholine receptor antagonist that activates proopiomelanocortin (POMC) neurons concentrated in the hypothalamus. The combination of these two therapies causes loss of appetite with increased energy. Naltrexone is a pure opioid antagonist, further enhancing POMC neuron activation and promoting a synergistic effect that helps keep cravings at bay.
While the solution to obesity relies largely on major lifestyle and nutrition modifications, weight loss surgery and long-term pharmacologic options are steadily growing in popularity among patients who, despite more conservative interventions and efforts, can’t seem to lose weight. In terms of pharmacologic interventions, it is only recently that these agents have begun to address the underlying behavioral drive of eating.
RELATED : ORBERA Intragastric Balloon Non-Surgical Weight Loss Device Receives FDA Approval
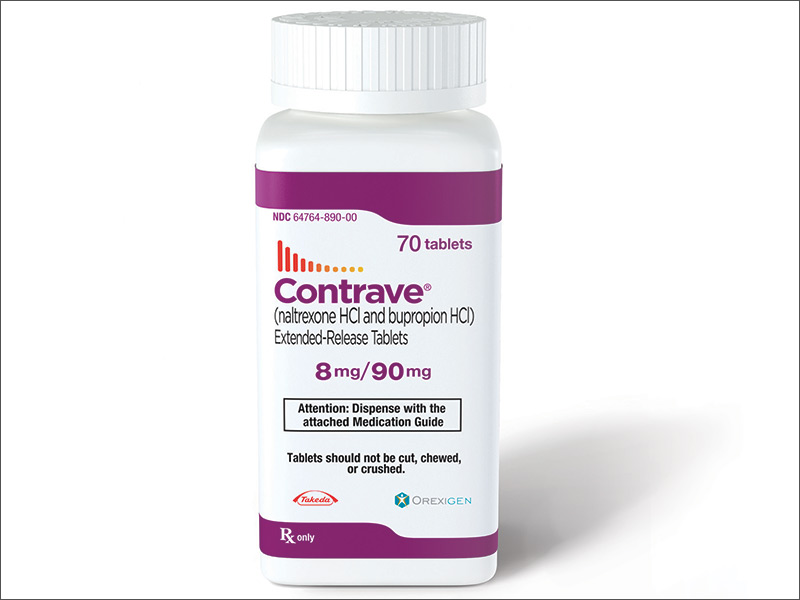
A sustained release formulation of naltrexone/bupropion was approved in the United States in September 2014 under the brand name Contrave, followed by a Spring 2015 approval in Europe under the name Mysimba. This combination operates on both the hypothalamic melancortin and the reward system in the brain, effectively demonstrating in clinical trials an ability to sustain clinically significant weight loss in obese patients and promote cardiovascular health.
About Contrave
Contrave is a trademark of Orexigen Therapeutics, Inc., and licensed by Takeda Pharmaceuticals America, Inc. It is comprised of 8mg of Naltrexone HCI and 90 mg of Bupropion. Today, Contrave is only available in tablet form and should be taken according to a physician’s instructions. The drug developer recommends taking one pill in the morning during the first week, one in the morning and another in the evening during the second week, the addition of another pill in the morning at third week, and four pills divided equally into the morning and evening during the following weeks.